For assistance in registering for an appointment, contact the Citizen Service Center at (626) 744-7311. Register for an appointment on MyTurn.CA.gov. The Pasadena Public Health Department (PPHD) is now administering first and second booster doses. Learn more about the updated guidance on the use of Janssen (Johnson & Johnson) COVID-19 vaccine Individuals ages 50 years and older or ages 12 and older who are moderately and severely immunocompromised are eligible to receive a second booster 4 months after the first boosterĬDC has updated its recommendations for COVID-19 vaccines with a preference for mRNA (Pfizer-BioNTech or Moderna) vaccines. Everyone ages 5 years and older who received their second dose of Pfizer or Moderna vaccine five months ago or ages 18 and older the Johnson & Johnson vaccine two months ago can get the booster of their choice. The Centers for Disease Control and Prevention (CDC) and the California Department of Public Health recommend a booster dose for fully vaccinated adults. An additional vaccine dose is not currently recommended by the CDC for immunocompromised persons who received a single dose of the J&J vaccine.
#COVID 19 VACCINE NEAR ME SERIES#
The Centers for Disease Control and Prevention (CDC) recommends an additional mRNA COVID-19 vaccine dose (Pfizer-BioNTech or Moderna) for moderately and severely immunocompromised people after an initial 2-dose primary mRNA vaccine series at least 4 weeks (28 days), because people with compromised immune systems may have a reduced ability to respond to vaccines, including COVID-19. The Pfizer vaccine also continues to be available under emergency use authorization (EUA), including for individuals 6 months through 15 years of age. The vaccine has been known as the Pfizer-BioNTech COVID-19 Vaccine, and will now be marketed as Comirnaty, for the prevention of COVID-19 disease in individuals 16 years of age and older. On August 23, 2021, the FDA approved the first COVID-19 vaccine.
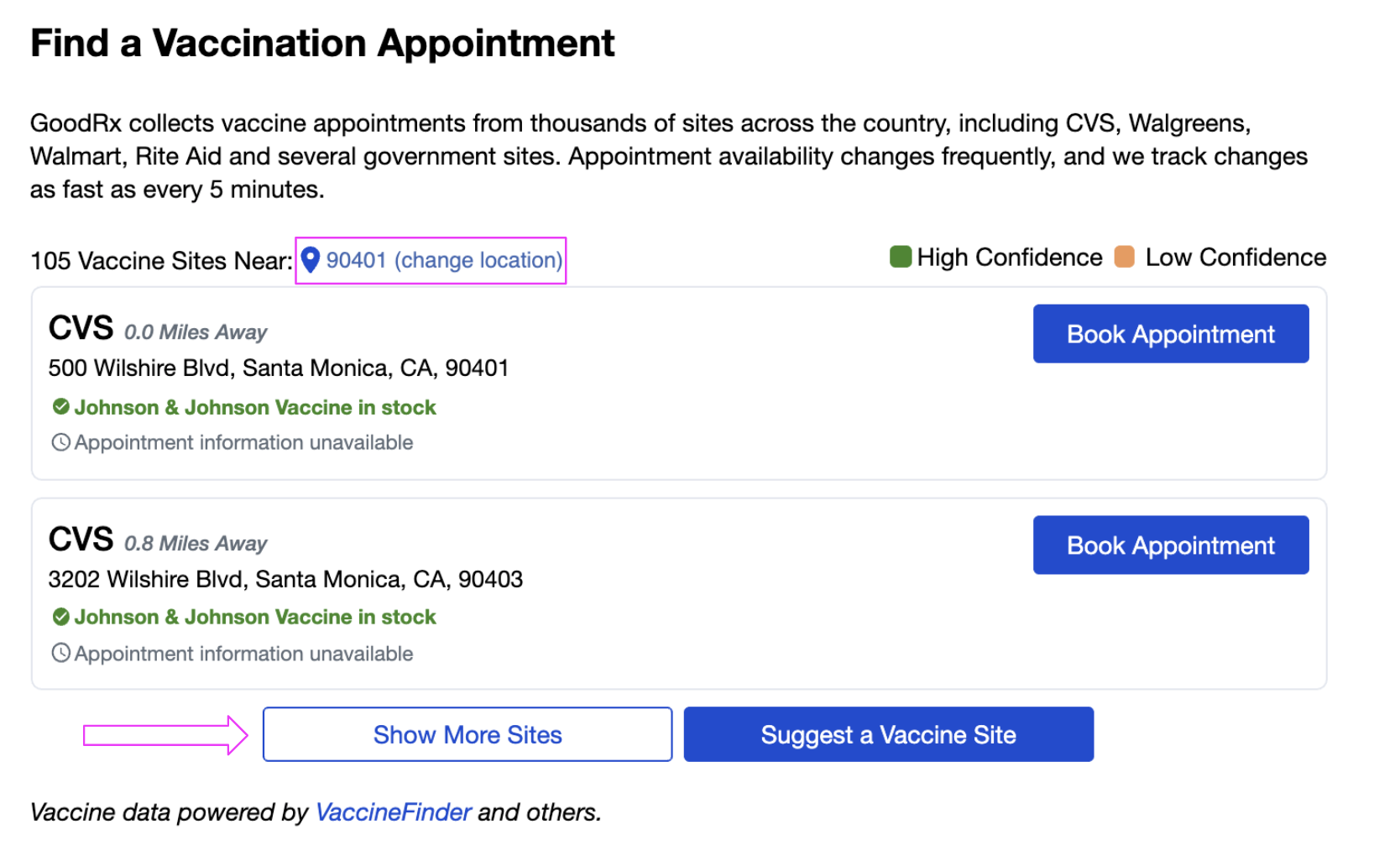
The vaccines are reactogenic, which means they can cause sore arm, fatigue, headache, and even low-grade fever that lasts one or two days. All vaccines are highly effective at preventing disease. Three COVID-19 vaccines have received emergency authorization for use in the U.S. Food and Drug Administration (FDA) or recommended for widespread use. are required to go through extensive safety testing before they are licensed by the U.S. That continues to be true when it comes to vaccines.Īll vaccines used in the U.S.

Home Instruction for Parents of Preschool Youngsters (HIPPY)įrom the start of the pandemic, data and science have guided Pasadena’s response to COVID-19.Maternal, Child, and Adolescent Health Program.Communicable Disease Prevention / Public Health Nursing.Women, Infants, and Children (WIC) Program.Health Promotion and Policy Development.
